- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
Mestag Therapeutics to Present Targeted Cancer Therapy at AACR
Biotech company selected for late-breaking poster on FAP-targeted LTBR agonist MST-0312 at major oncology conference.
Apr. 14, 2026 at 7:49am
Got story updates? Submit your updates here. ›
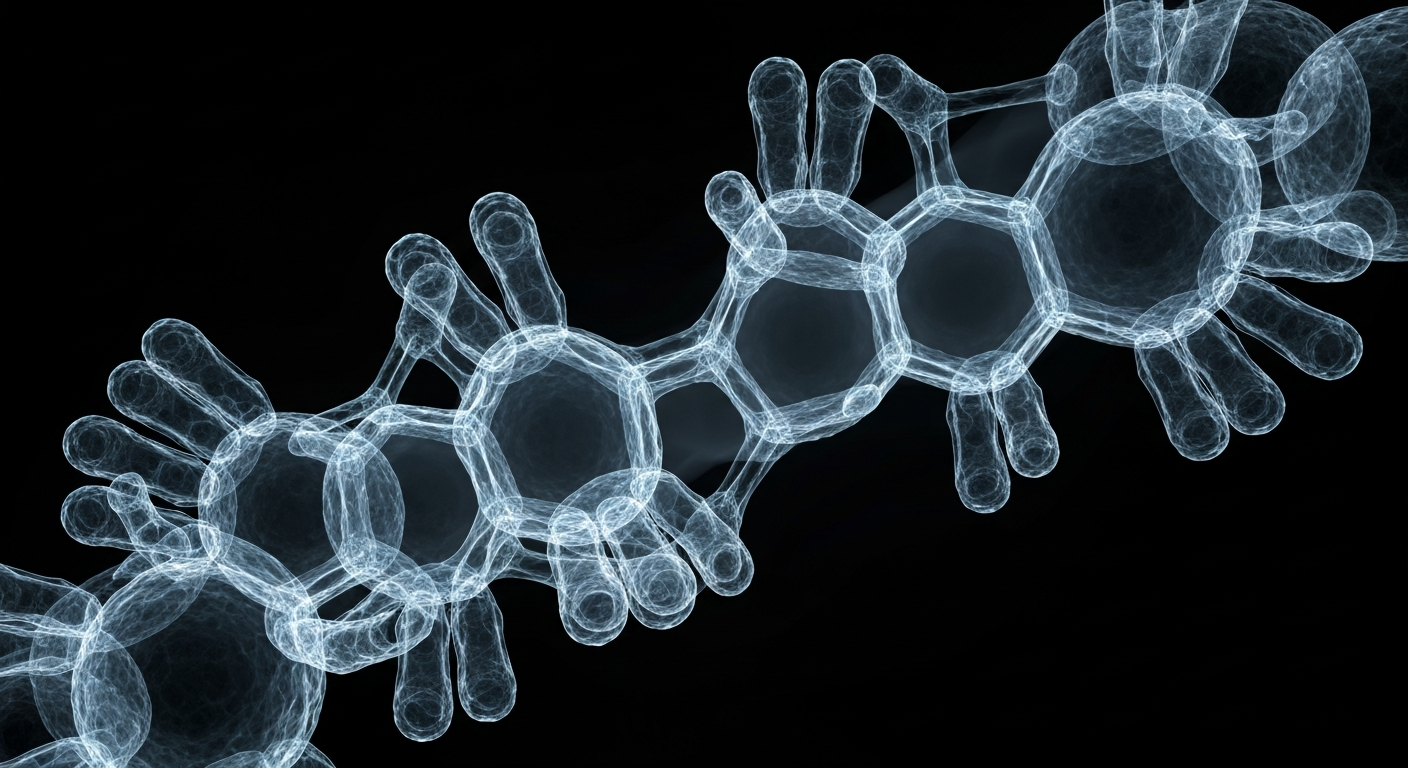 A novel bispecific antibody designed to harness the power of the immune system against solid tumors.San Diego Today
A novel bispecific antibody designed to harness the power of the immune system against solid tumors.San Diego TodayMestag Therapeutics, a biotech company focused on fibroblast-based immunotherapies, announced it has been selected to present pre-clinical data on its FAP-targeted LTBR agonist antibody MST-0312 in a late-breaking poster session at the upcoming American Association for Cancer Research (AACR) Annual Meeting. MST-0312 is designed to induce the formation of tertiary lymphoid structures and high endothelial venules in solid tumors, which can enhance immune cell access and activity within the tumor microenvironment.
Why it matters
The presence of tertiary lymphoid structures and high endothelial venules in solid tumors has been associated with improved patient survival and better responses to cancer immunotherapies. Mestag's approach aims to leverage this fibroblast-immune biology to develop a new class of targeted cancer treatments.
The details
MST-0312 is a bispecific antibody that binds to fibroblast activation protein (FAP) on tumor-associated fibroblasts and activates the lymphotoxin beta receptor (LTBR) pathway. This activation is intended to induce the formation of tertiary lymphoid structures and high endothelial venules within the tumor, which can facilitate greater infiltration and activity of anti-tumor immune cells. Mestag plans to initiate a Phase 1 clinical trial of MST-0312, called the STARLYS trial, in mid-2026.
- The AACR Annual Meeting will take place from April 17-22, 2026 in San Diego, California.
- Mestag's late-breaking poster presentation on MST-0312 is scheduled for Tuesday, April 21, 2026 from 9:00am to 12:00pm.
- Mestag plans to initiate the Phase 1 STARLYS trial for MST-0312 in mid-2026.
The players
Mestag Therapeutics
A biotech company harnessing fibroblast immunology to develop treatments for cancer and inflammatory diseases.
Pascal Merchiers
Chief Development Officer at Mestag Therapeutics, who will present the data on MST-0312 at the AACR Annual Meeting.
American Association for Cancer Research (AACR)
A major oncology conference where Mestag has been selected to present its late-breaking research.
What they’re saying
“MST-0312 is a FAP-targeted LTBR agonist bispecific antibody designed to induce the formation of tertiary lymphoid structures (TLS) and high endothelial venules (HEV) in solid tumors.”
— Pascal Merchiers, Chief Development Officer, Mestag Therapeutics
What’s next
Mestag plans to initiate the Phase 1 STARLYS trial for MST-0312 in mid-2026 to further evaluate the safety and efficacy of the targeted LTBR agonist in solid tumor patients.
The takeaway
Mestag's approach to leveraging fibroblast-immune biology represents a novel therapeutic strategy in oncology, with the potential to enhance immune cell infiltration and activity within solid tumors. The upcoming AACR presentation will provide an important first look at the preclinical data supporting this targeted LTBR agonist program.
San Diego top stories
San Diego events
Apr. 14, 2026
2026 Aztec Baseball vs. UC San DiegoApr. 14, 2026
San Diego Padres vs. Seattle MarinersApr. 14, 2026
Ethel Cain - The Willoughby Tucker Forever Tour




