- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
Brain Pacemaker Offers New Hope for Parkinson's Patients
Adaptive deep brain stimulation (aDBS) device transforms the lives of those living with Parkinson's disease.
Apr. 12, 2026 at 11:04am
Got story updates? Submit your updates here. ›
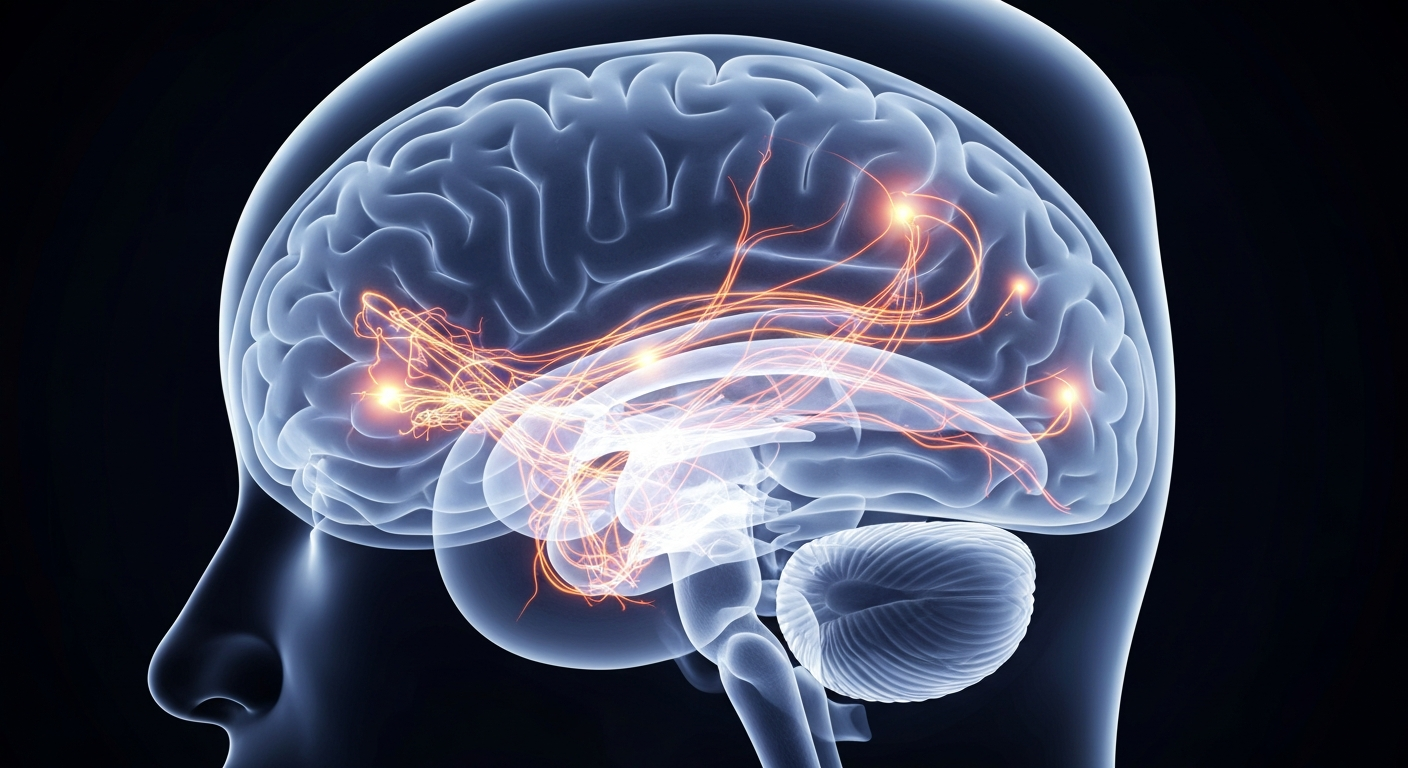 A revolutionary brain pacemaker is giving new hope to Parkinson's patients by adapting to their individual brain activity.Stanford Today
A revolutionary brain pacemaker is giving new hope to Parkinson's patients by adapting to their individual brain activity.Stanford TodayA revolutionary brain pacemaker called adaptive deep brain stimulation (aDBS) is giving new hope to Parkinson's patients. The device, recently approved by the FDA, senses brain activity in real-time and delivers electrical pulses only when needed, unlike traditional deep brain stimulation which provides constant stimulation. Stanford professor Keith Krehbiel, who has lived with Parkinson's for decades, was the first patient to receive the aDBS device and has experienced life-changing improvements in his symptoms.
Why it matters
Parkinson's disease affects an estimated 1.1 million Americans, causing debilitating symptoms like tremors, stiffness, and balance issues. The aDBS device represents a major breakthrough in Parkinson's treatment, offering a more personalized and effective therapy that could dramatically improve quality of life for those living with the disease.
The details
The aDBS device works like a pacemaker for the brain, constantly monitoring brain activity and adjusting electrical stimulation accordingly. This is a significant improvement over traditional deep brain stimulation, which delivers a constant train of electrical pulses. Dr. Helen Bronte-Stewart, a Stanford professor who has pioneered this technology, explains that the adaptive nature of aDBS allows it to 'listen' to the brain and respond in real-time, providing more targeted and effective treatment.
- In the summer of 2020, Keith Krehbiel became the first patient to receive the aDBS device as part of a groundbreaking international clinical trial.
- The aDBS device was approved by the FDA in February 2025 and was named one of Time magazine's 'Best Inventions of 2025'.
The players
Keith Krehbiel
A Stanford professor who has been living with Parkinson's disease for nearly three decades and was the first patient to receive the aDBS device.
Dr. Helen Bronte-Stewart
A Stanford Medicine professor who has dedicated her career to understanding how the brain controls movement and finding ways to correct disorders, leading to the development and FDA approval of the aDBS device.
Medtronic
The company that manufactures the aDBS device, which was named one of Time magazine's 'Best Inventions of 2025'.
What they’re saying
“Falling is a common symptom of people with Parkinson's.”
— Keith Krehbiel, Stanford professor and Parkinson's patient
“Until recently, these stimulation devices delivered a one-size-fits-all train of electric pulses to the brain around the clock. Now, we have this adaptive technology that listens to brain activity and adjusts stimulation accordingly.”
— Dr. Helen Bronte-Stewart, Stanford Medicine professor
“When it's on and working, then people really do [improve]. What they say is their disease goes back at least five years.”
— Dr. Helen Bronte-Stewart, Stanford Medicine professor
What’s next
Researchers and medical professionals will continue to study the long-term effects of the aDBS device and explore ways to further refine and improve this revolutionary Parkinson's treatment.
The takeaway
The story of Keith Krehbiel and the aDBS device highlights the transformative potential of innovative technology to improve the lives of those living with Parkinson's disease. As medical science continues to push the boundaries of what's possible, the future of Parkinson's treatment looks brighter than ever.
Stanford top stories
Stanford events
Apr. 16, 2026
David ByrneApr. 17, 2026
Stanford Cardinal Baseball vs. Miami Hurricanes Baseball




