- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
Experimental Therapy Helps Liver Transplant Patients Avoid Anti-Rejection Drugs
A small clinical trial shows some liver transplant recipients can remain drug-free for at least 3 years after novel cell therapy.
Apr. 18, 2026 at 1:59pm
Got story updates? Submit your updates here. ›
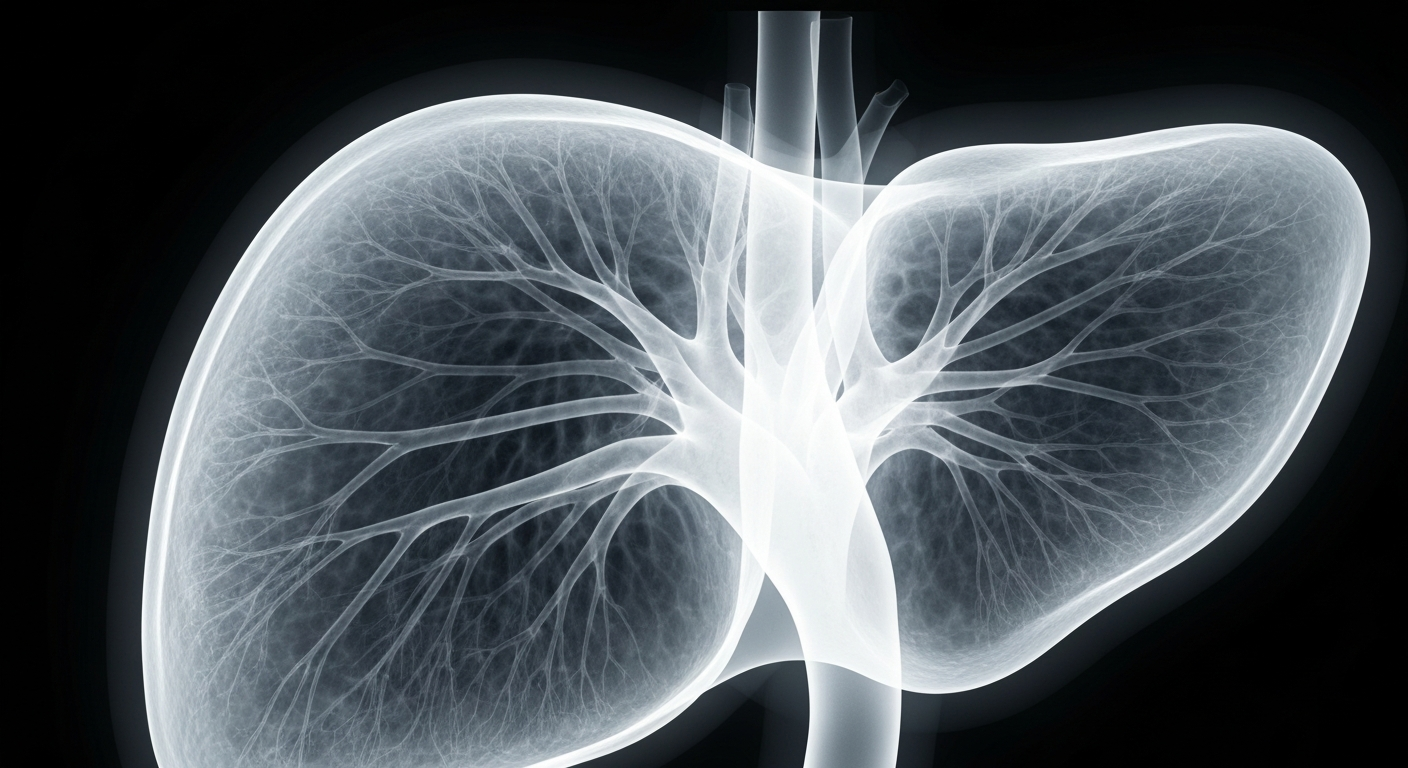 Experimental cell therapy aims to retrain the immune system to accept transplanted livers without the need for lifelong anti-rejection drugs.Today in Pittsburgh
Experimental cell therapy aims to retrain the immune system to accept transplanted livers without the need for lifelong anti-rejection drugs.Today in PittsburghA small but significant clinical trial has demonstrated that some liver transplant recipients can remain free of immunosuppressive drugs for at least three years following a novel experimental therapy involving infusion of regulatory T cells harvested from the donor. Of the 12 patients in the study, 8 were successfully weaned off all anti-rejection medications within 6 months to a year post-transplant, with 6 of those remaining drug-free for at least 3 years.
Why it matters
Immunosuppressive drugs carry significant long-term risks for transplant recipients, including kidney damage, increased susceptibility to infections, and higher rates of malignancy. Eliminating or reducing dependence on these medications could substantially improve quality of life and long-term survival for transplant patients.
The details
The findings, reported by researchers at the University of Pittsburgh Medical Center and published in the journal Hepatology, stem from a phase 1/2 study involving 12 patients who received liver transplants between 2019 and 2021. All participants underwent a standard deceased-donor liver transplant followed by an infusion of regulatory T cells (Tregs) harvested from the donor and expanded ex vivo. The goal was to induce immune tolerance, thereby reducing or eliminating the need for lifelong immunosuppression.
- The study involved patients who received liver transplants between 2019 and 2021.
- As of the most recent follow-up, six of the eight patients who were successfully weaned off anti-rejection drugs have remained drug-free for at least three years.
The players
University of Pittsburgh Medical Center
The research institution where the clinical trial was conducted and the findings were reported.
Angus Thomson
The senior author of the study and director of transplant immunology at the University of Pittsburgh.
Hepatology
The academic journal in which the study was published.
What they’re saying
“We're seeing durable tolerance in a subset of patients, which suggests that cellular therapy can play a role in reprogramming the immune response after transplantation.”
— Angus Thomson, Director of transplant immunology, University of Pittsburgh
What’s next
The research team is now preparing for a multi-center phase 2 trial that will incorporate randomized controls and expanded biomarker analysis to better predict treatment response. Funding for the next phase is being pursued through the National Institutes of Health's Human Immunology Project Consortium, with preliminary discussions underway with cellular therapy manufacturers to scale Treg production under good manufacturing practice (GMP) standards.
The takeaway
If future trials confirm these results, Treg therapy could represent a paradigm shift in transplant care — moving from lifelong pharmacological management toward immune system retraining. For now, the approach remains investigational, available only through clinical trials, and not yet approved by regulatory agencies such as the FDA or EMA.
Pittsburgh top stories
Pittsburgh events
Apr. 19, 2026
Pittsburgh Pirates vs. Tampa Bay RaysApr. 19, 2026
Mama Said String BandApr. 19, 2026
Hamilton Sing-Along Drag Brunch




