- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
Radical Catheter Technologies Announces FDA Clearance for 6F Neurovascular Catheter
New device broadens access to advanced neurovascular procedures across radial and femoral approaches.
Apr. 14, 2026 at 6:14am
Got story updates? Submit your updates here. ›
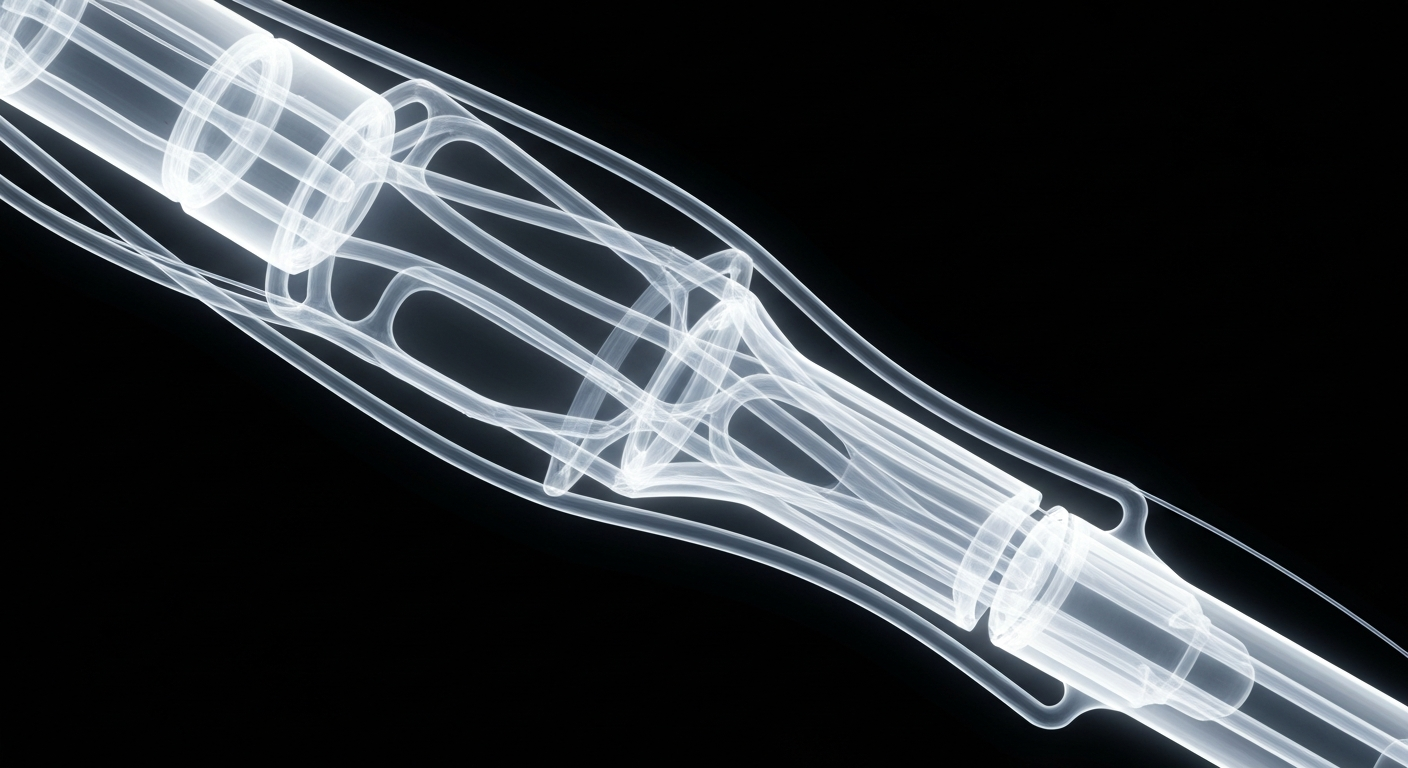 Radical Catheter Technologies' new 6F neurovascular device, revealed through the ghostly, translucent lines of an X-ray photograph, showcases the innovation and technical sophistication behind this breakthrough endovascular technology.NYC Today
Radical Catheter Technologies' new 6F neurovascular device, revealed through the ghostly, translucent lines of an X-ray photograph, showcases the innovation and technical sophistication behind this breakthrough endovascular technology.NYC TodayRadical Catheter Technologies, a portfolio company of NeuroTechnology Investors (NTI), announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for its 6F neurovascular access catheter. This latest addition to Radical's patented catheter platform expands therapeutic options, particularly for middle-meningeal artery (MMA) embolization, and further increases availability in radial access procedures.
Why it matters
The FDA clearance of Radical's 6F catheter represents an important advancement in neurovascular care, providing physicians with a best-in-class device that delivers improved performance and flexibility across a wide range of complex endovascular procedures. This innovation aligns with Radical's mission to address longstanding gaps in neurovascular device technology and support the rollout of other transformative neurological solutions through its specialized commercialization team.
The details
Radical's 6F catheter is built on patented ribbon technology designed to offer simultaneous flexibility, pushability, and stability, along with the durability physicians need when navigating challenging anatomy. The device's thinner wall construction provides a greater inner diameter relative to outer diameter, and a smooth, continuous liner enables the delivery of other therapeutic devices. These mechanical advantages are intended to enhance procedural efficiency and performance.
- Radical Catheter Technologies received FDA 510(k) clearance for the 6F neurovascular catheter on April 13, 2026.
- The company plans to exhibit its patented technology platform at the Society of Neurointerventional Science's (SNIS) 23rd Annual Meeting from July 20-24, 2026, in Seattle, Washington.
The players
Radical Catheter Technologies
A pioneering medical device company advancing a patented catheter platform designed to support next-generation endovascular access and delivery.
NeuroTechnology Investors (NTI)
A leading investment group dedicated to advancing breakthrough neurological technologies in the medical device sector, with a diverse portfolio that includes Radical Catheter Technologies.
Johanna Fifi, MD
A neurovascular surgeon and Professor of Neurosurgery, Neurology, and Radiology at The Mount Sinai Hospital in New York.
Brian Martin
The CEO and co-founder of Radical Catheter Technologies.
Alex Thomson
The Vice President of Commercial at Radical Catheter Technologies.
What they’re saying
“The FDA clearance of the 6F Radical catheter is a meaningful advancement for our patients. The 6F's lower profile delivers best-in-class performance across both radial and femoral access. Built on the foundation Radical Catheter Technologies has already established, the 6F becomes an immediate top choice, particularly in challenging cases.”
— Johanna Fifi, MD, Neurovascular surgeon and Professor of Neurosurgery, Neurology, and Radiology at The Mount Sinai Hospital, New York
“We have spent years developing a truly innovative, best-in-class catheter platform that directly addresses performance gaps physicians have long identified. With our novel technology, we are exceeding these expectations and positioning the Radical platform to serve as the foundation for continued innovation in neuroendovascular procedures.”
— Brian Martin, CEO and co-founder of Radical Catheter Technologies
“The FDA 510(k) clearance of our latest 6F access catheter further validates the breadth of our platform across a wide range of neurovascular applications. This commercialization model unites some of the most compelling technologies in the neurovascular space, while providing the infrastructure to advance and support the best-in-class solutions we represent.”
— Alex Thomson, Vice President of Commercial at Radical Catheter Technologies
What’s next
Radical Catheter Technologies plans to exhibit its patented technology platform, including the new 6F neurovascular catheter, at the Society of Neurointerventional Science's (SNIS) 23rd Annual Meeting in Seattle, Washington, from July 20-24, 2026.
The takeaway
The FDA clearance of Radical's 6F neurovascular catheter represents a significant advancement in endovascular device technology, providing physicians with a best-in-class solution that enhances procedural performance and flexibility. This innovation aligns with Radical's mission to address longstanding gaps in neurovascular care and support the rollout of transformative neurological solutions through its specialized commercialization team.





