- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
Emalex Receives U.S. Patent for Orally Disintegrating Ecopipam Formulation
The new drug formulation could provide a more convenient treatment option.
Apr. 9, 2026 at 4:37pm
Got story updates? Submit your updates here. ›
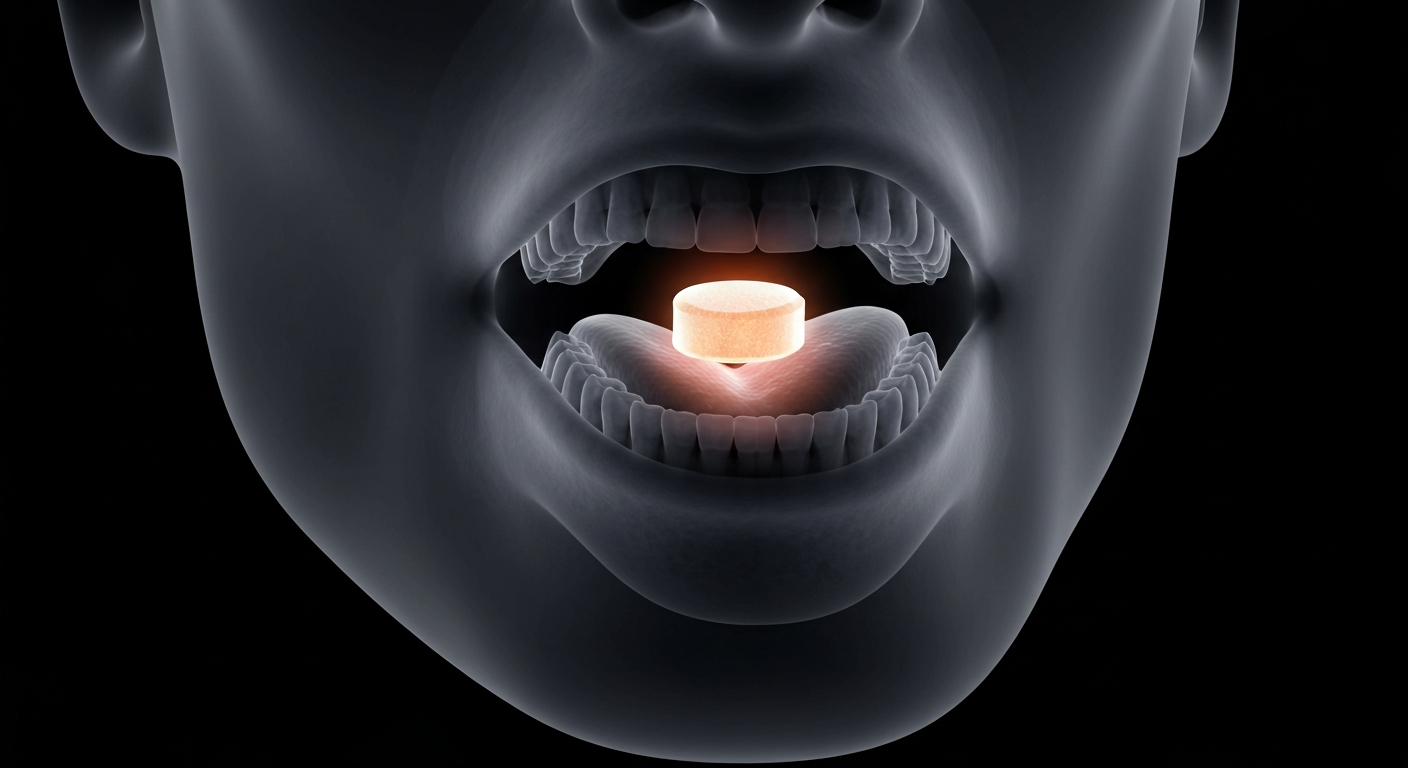 A new orally disintegrating drug formulation aims to make a neurological treatment more accessible and convenient for patients.Chicago Today
A new orally disintegrating drug formulation aims to make a neurological treatment more accessible and convenient for patients.Chicago TodayEmalex, a pharmaceutical company, has received a U.S. patent for an orally disintegrating formulation of their drug ecopipam. This new formulation is designed to dissolve quickly in the mouth, potentially making it easier for patients to take the medication.
Why it matters
Ecopipam is used to treat certain neurological and psychiatric disorders. An orally disintegrating formulation could improve patient compliance and access to the drug, as it may be easier for some patients to take compared to traditional tablet or capsule forms.
The details
The patented formulation uses proprietary technology to create an orally disintegrating tablet that dissolves rapidly in the mouth. This could provide a more convenient treatment option for patients who have difficulty swallowing traditional solid oral dosage forms.
- Emalex received the U.S. patent for the orally disintegrating ecopipam formulation on April 9, 2026.
The players
Emalex
A pharmaceutical company that develops treatments for neurological and psychiatric disorders.
Ecopipam
A drug developed by Emalex to treat certain neurological and psychiatric conditions.
What’s next
Emalex plans to further develop the orally disintegrating ecopipam formulation and seek regulatory approval for its use.
The takeaway
This new orally disintegrating formulation of ecopipam could improve patient access and compliance for those seeking treatment for neurological or psychiatric disorders, providing a more convenient dosage option.
Chicago top stories
Chicago events
Apr. 15, 2026
Hamilton (Chicago)Apr. 15, 2026
The Merry Wives of WindsorApr. 15, 2026
Hamilton (Chicago)




