- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
$3 Million Prize Awarded for Research Leading to First Sickle Cell CRISPR Therapy
Two scientists recognized for groundbreaking work on gene-editing treatment for blood disorders.
Apr. 19, 2026 at 12:03am
Got story updates? Submit your updates here. ›
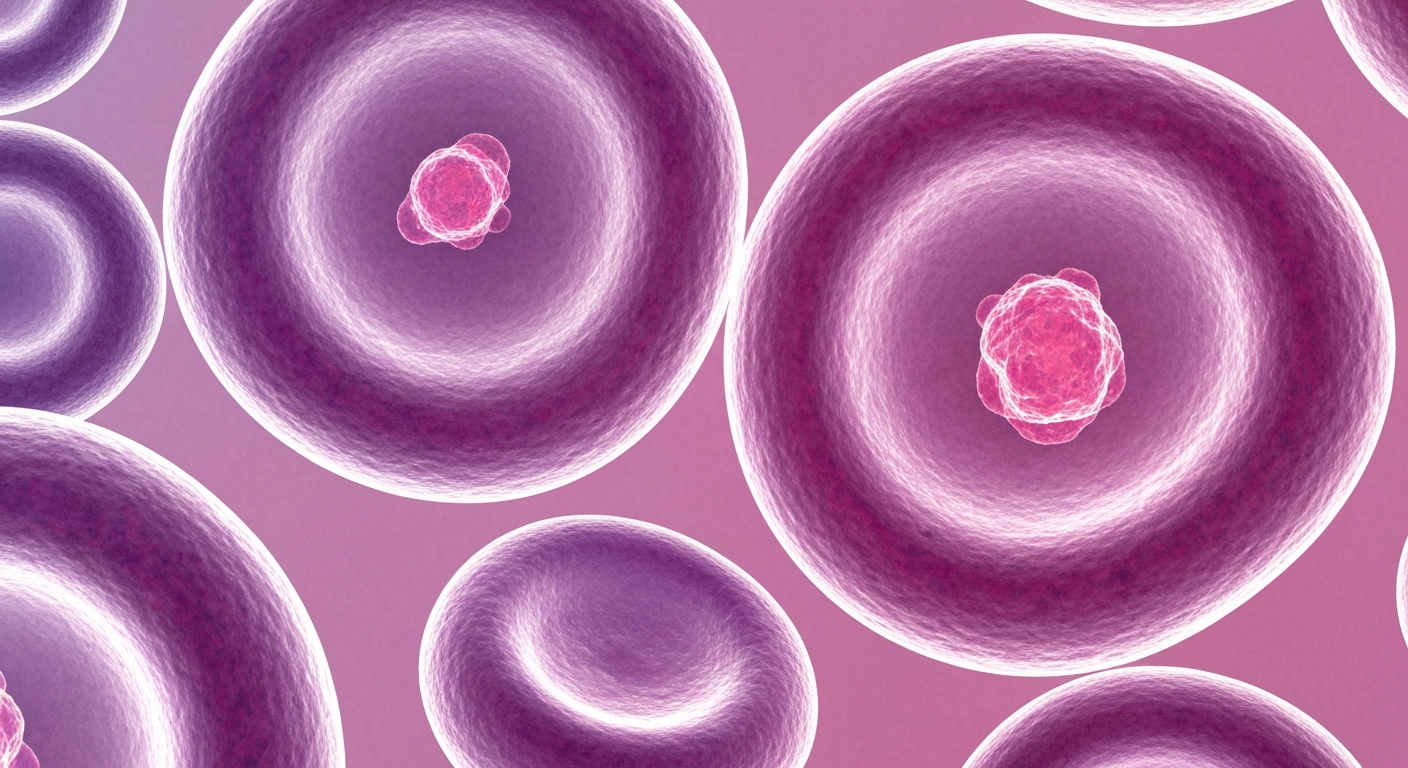 Groundbreaking gene therapy research has led to a new CRISPR-based treatment that can functionally cure devastating blood disorders by targeting the genetic root cause.Los Angeles Today
Groundbreaking gene therapy research has led to a new CRISPR-based treatment that can functionally cure devastating blood disorders by targeting the genetic root cause.Los Angeles TodayTwo researchers, Dr. Swee Lay Thein of the National Heart, Lung and Blood Institute and Dr. Stuart H. Orkin of Harvard University, have been awarded the $3 million Breakthrough Prize in Life Sciences for their work that led to the development of the first approved gene therapy using CRISPR to treat sickle cell disease and beta-thalassemia. The treatment, called Casgevy, functionally cures patients of these painful and potentially deadly blood disorders by disabling a single gene.
Why it matters
Sickle cell disease affects millions globally, predominantly in Africa, and causes debilitating pain crises and organ damage. Beta-thalassemia also severely impacts quality of life, requiring lifelong blood transfusions. The Casgevy therapy represents a major breakthrough in treating these previously incurable conditions, but access and affordability remain significant challenges, especially in underserved regions.
The details
Thein's research in the 1980s uncovered how certain genetic variants allow continued production of fetal hemoglobin, which can mitigate sickle cell symptoms. Orkin's work then showed that the BCL11A gene acts as a 'repressor' that turns off fetal hemoglobin production. By using CRISPR to disable BCL11A, the Casgevy therapy is able to reactivate fetal hemoglobin and effectively cure patients. However, the complex and costly process of extracting, editing, and reinfusing a patient's bone marrow cells limits accessibility, leading researchers to explore simpler 'in vivo' CRISPR approaches.
- The Breakthrough Prize ceremony was held on April 18, 2026 in Los Angeles.
- Thein began her research on sickle cell and beta-thalassemia in the 1980s.
The players
Dr. Swee Lay Thein
A Malaysian hematologist and physician at the National Heart, Lung and Blood Institute (NHLBI) who co-won the 2026 Breakthrough Prize for her pioneering research on sickle cell and beta-thalassemia.
Dr. Stuart H. Orkin
A pediatric hematologist and oncologist at Boston Children's Hospital, Dana-Farber Cancer Institute, Harvard Medical School, and Howard Hughes Medical Institute who co-won the 2026 Breakthrough Prize for his research showing BCL11A as a viable target for gene therapy.
Casgevy
The first approved gene therapy using CRISPR to treat sickle cell disease and beta-thalassemia by disabling the BCL11A gene to reactivate fetal hemoglobin production.
What they’re saying
“I feel extremely honored, overwhelmed and humbled.”
— Dr. Swee Lay Thein
What’s next
Researchers are now pivoting to explore 'in vivo' CRISPR approaches that involve directly injecting the gene-editing machinery into patients, which could make the treatment more accessible and affordable compared to the current complex process of extracting, editing, and reinfusing bone marrow cells.
The takeaway
The Casgevy gene therapy represents a major breakthrough in treating previously incurable blood disorders like sickle cell disease and beta-thalassemia, but significant challenges remain in making this cutting-edge treatment widely available, especially in underserved regions where these conditions are most prevalent.
Los Angeles top stories
Los Angeles events
Apr. 19, 2026
TINA - The Tina Turner Musical (touring)Apr. 19, 2026
TINA - The Tina Turner Musical (touring)Apr. 19, 2026
Kim's Convenience




