- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
Gene Editing Therapy Shows Promise for Severe Sickle Cell
Clinical trial results indicate a one-time gene-edited treatment could provide a 'functional cure' for the debilitating blood disorder.
Apr. 2, 2026 at 8:03am
Got story updates? Submit your updates here. ›
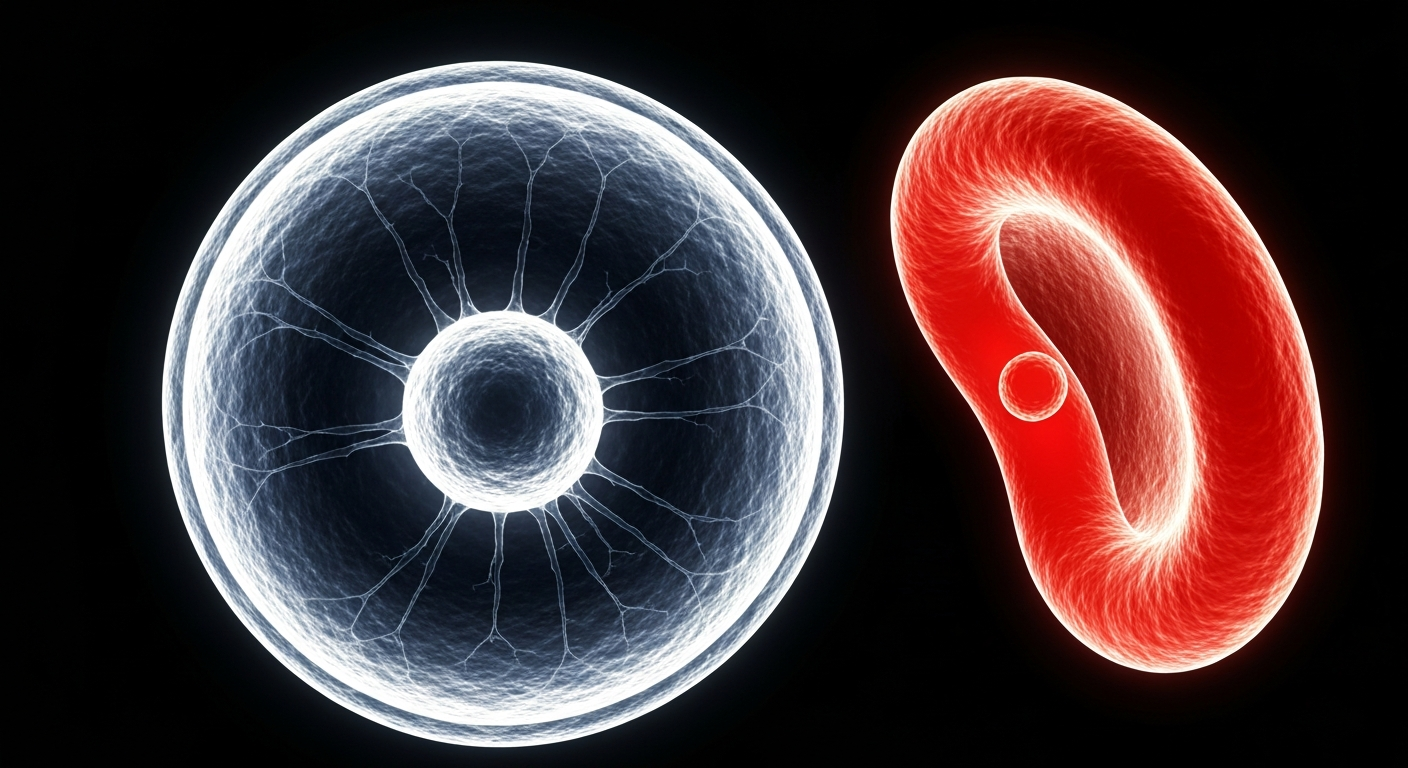 Groundbreaking gene-editing therapy aims to correct the genetic mutation that causes sickle-shaped red blood cells, a debilitating condition with few curative options.Cleveland Today
Groundbreaking gene-editing therapy aims to correct the genetic mutation that causes sickle-shaped red blood cells, a debilitating condition with few curative options.Cleveland TodayA new gene-editing cell therapy has shown promising results in a clinical trial for treating severe sickle cell disease, a genetic blood disorder with limited curative options. The experimental treatment, called Renizgamglogene autogedtemcel (reni-cel), was able to increase levels of fetal hemoglobin and improve overall hemoglobin levels in 27 out of 28 patients, effectively preventing painful sickle cell crises.
Why it matters
Sickle cell disease is a debilitating condition that can lead to severe pain, organ damage, and a shortened lifespan. Current treatments, including blood or marrow transplants, carry significant risks. This new gene-editing therapy offers hope for a potential 'functional cure' that could dramatically improve quality of life for those living with severe sickle cell disease.
The details
In the RUBY Trial, researchers used CRISPR/Cas12a gene-editing technology to modify patients' own blood-forming stem cells. This increased fetal hemoglobin levels, which prevents red blood cells from forming the sickle shape that causes complications. After treatment, 27 out of 28 patients did not experience any further sickle cell crises, and their average hemoglobin levels rose to near-normal levels.
- The RUBY Trial results were published in the New England Journal of Medicine in April 2026.
- The 28 patients, including 4 treated at Cleveland Clinic Children's, underwent the gene-editing procedure in 2025.
The players
Rabi Hanna, M.D.
Lead author of the RUBY Trial study and chair of the Pediatric Hematology – Oncology & Blood and Bone Marrow Transplant Division at Cleveland Clinic Children's.
Cleveland Clinic Children's
A specialized center for the care of adults and children with sickle cell disease, providing comprehensive treatment and support services.
Editas Medicine
The sponsor of the RUBY Trial, which is testing the gene-editing therapy Renizgamglogene autogedtemcel (reni-cel).
What they’re saying
“We have seen that a benefit of this CRISPR/Cas12a gene-editing technology is that there is no rejection, so it's different from traditional bone marrow transplants, which is standard treatment for sickle cell patients currently. Our aim has been to achieve a functional cure to help prevent any future damage caused by sickle cell disease, and these latest results are compelling.”
— Rabi Hanna, M.D., Lead author and chair of the Pediatric Hematology – Oncology & Blood and Bone Marrow Transplant Division at Cleveland Clinic Children's
What’s next
The RUBY Trial is ongoing, and researchers will continue to monitor the long-term safety and efficacy of the gene-editing therapy for sickle cell disease.
The takeaway
This promising gene-editing treatment represents a major advancement in the search for a cure for sickle cell disease, offering hope for a future where this debilitating condition can be effectively managed or even eliminated through innovative medical breakthroughs.
Cleveland top stories
Cleveland events
Apr. 2, 2026
Poison the Well "Peace in Place" TourApr. 2, 2026
Sparky a.k.a the Jukebox Troubadour




