- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
Tanner Pharma and Biodexa Launch Early Access Program for FAP Patients
The program will provide global access to eRapa, an investigational therapy for Familial Adenomatous Polyposis.
Mar. 31, 2026 at 2:08pm
Got story updates? Submit your updates here. ›
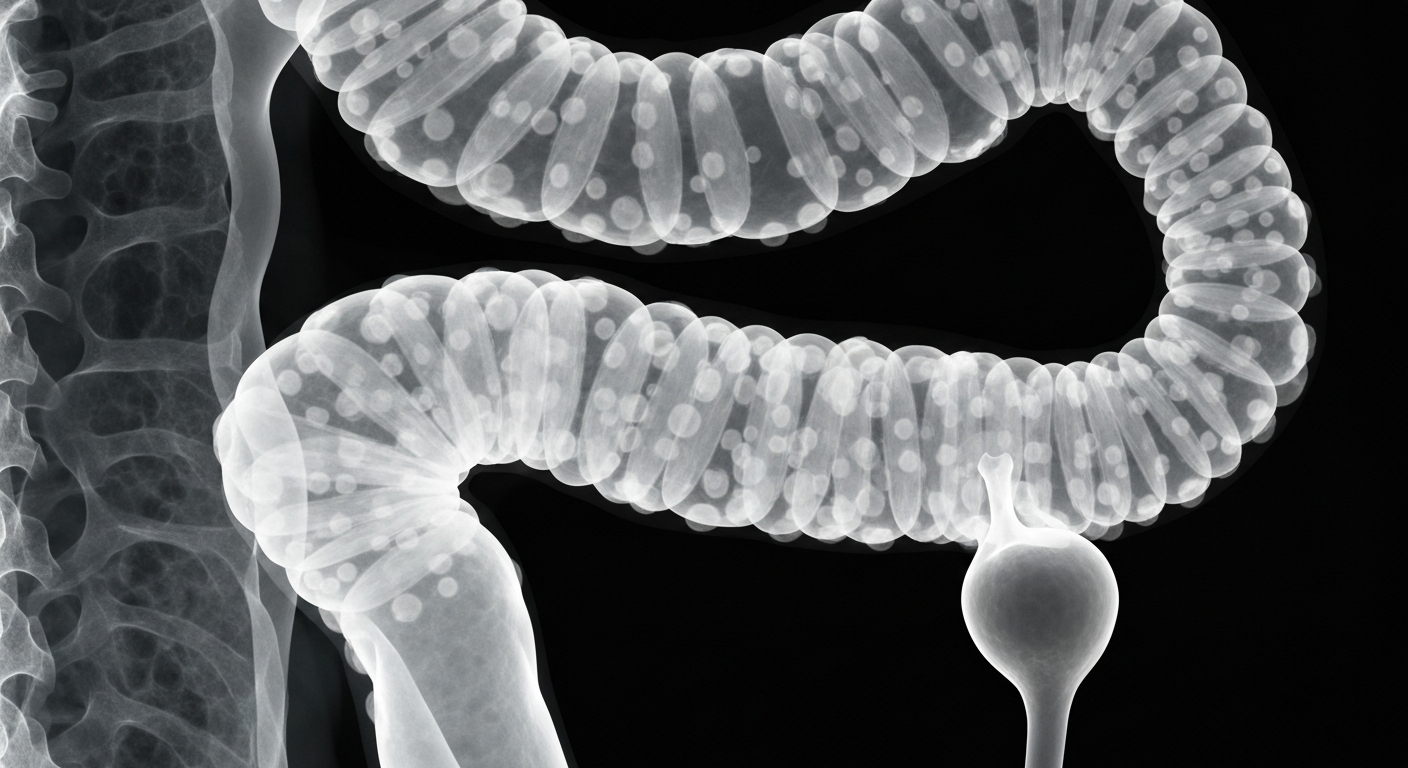 A new early access program aims to provide global access to an investigational therapy for Familial Adenomatous Polyposis, a rare genetic condition marked by the proliferation of polyps in the colon.Charlotte Today
A new early access program aims to provide global access to an investigational therapy for Familial Adenomatous Polyposis, a rare genetic condition marked by the proliferation of polyps in the colon.Charlotte TodayTanner Pharma Group, a global provider of specialty medicine access solutions, and Biodexa Pharmaceuticals, a clinical-stage biopharmaceutical company, have announced a strategic partnership to enable global access to eRapa, an investigational oral capsule formulation of rapamycin, for patients with Familial Adenomatous Polyposis (FAP) through an Early Access / Named Patient Program.
Why it matters
FAP is a rare genetic condition characterized by the development of numerous polyps in the colon and rectum, often leading to colorectal cancer if left untreated. Currently, there are no approved therapeutic options, and surgical resection remains the standard of care. This program aims to provide access to an investigational therapy that targets the underlying molecular mechanisms of FAP.
The details
Through this collaboration, Tanner Pharma will facilitate compliant, controlled access to eRapa in countries where it is not currently available, allowing clinicians treating FAP patients to prescribe this investigational therapy outside of a clinical trial for the first time. The program is designed to expand access while supporting real-world data collection to further advance the understanding of FAP and patient experience.
- The Early Access / Named Patient Program is launching in March 2026.
The players
Tanner Pharma Group
A global provider of specialty medicine access solutions, based in Charlotte, North Carolina, USA.
Biodexa Pharmaceuticals PLC
A clinical-stage biopharmaceutical company developing innovative products for gastrointestinal cancers, headquartered in Cardiff, UK.
eRapa
A proprietary oral capsule formulation of rapamycin (sirolimus), an mTOR inhibitor that has shown promise in treating FAP.
Familial Adenomatous Polyposis (FAP)
A rare genetic condition characterized by the development of numerous polyps in the colon and rectum, often leading to colorectal cancer if left untreated.
What’s next
As part of this initiative, Biodexa will also generate Real World Data (RWD) to further advance understanding of FAP and patient experience. Access through the program is based on patient eligibility, local regulations, and availability of government or private funding.
The takeaway
This early access program represents a significant step forward in providing treatment options for patients with Familial Adenomatous Polyposis, a rare and debilitating condition that currently lacks approved therapeutic options. By expanding access to an investigational therapy and collecting real-world data, this collaboration aims to improve the lives of FAP patients globally.
Charlotte top stories
Charlotte events
Apr. 2, 2026
Charlotte Hornets vs. Phoenix SunsApr. 7, 2026
Ali Wong Live



