- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
Waltham Today
By the People, for the People
Fortitude Biomedicines Announces Lead Program for Axial Spondyloarthritis
New T-cell targeting bispecific antibody enters clinical development
Apr. 7, 2026 at 2:39pm
Got story updates? Submit your updates here. ›
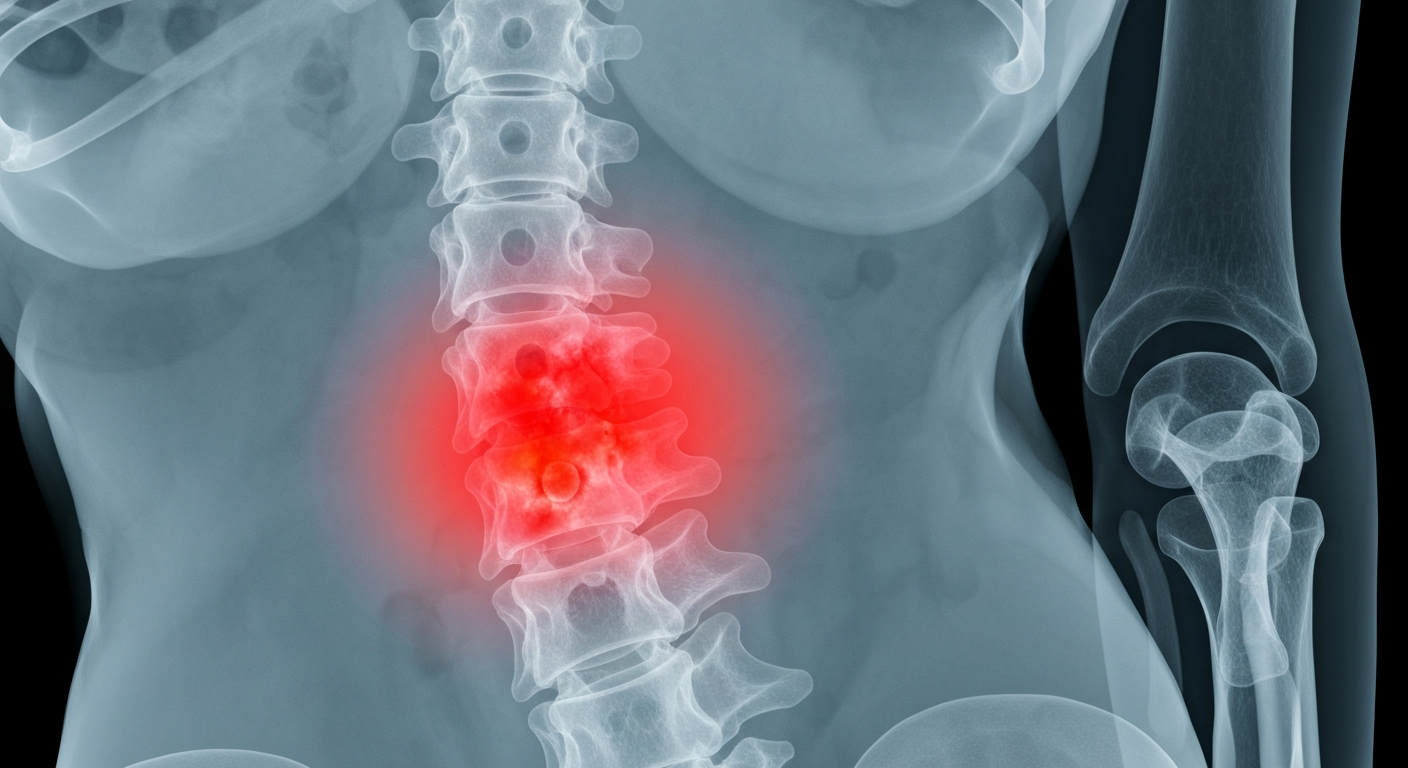 A revealing X-ray view of the internal damage caused by the chronic inflammatory condition of axial spondyloarthritis.Waltham Today
A revealing X-ray view of the internal damage caused by the chronic inflammatory condition of axial spondyloarthritis.Waltham TodayFortitude Biomedicines, a biopharmaceutical company focused on immune cell targeting biologics and antibody-drug conjugates, announced that its lead program is a first-in-class T-cell targeting bispecific antibody for the potential treatment of Axial Spondyloarthritis (AxSpA). The company also appointed Dr. Rahul Patel as Senior Vice President of Clinical Development to accelerate the clinical development of this program.
Why it matters
AxSpA is a chronic, debilitating inflammatory disease affecting the axial skeleton that impacts over 1.8 million people in the US and 50 million globally. Without effective treatment, AxSpA can lead to progressive structural damage and long-term functional impairment, significantly diminishing patients' quality of life. Fortitude's new program aims to address this large unmet medical need.
The details
Fortitude's lead AxSpA program has shown promising preclinical activity, with selective and durable suppression of disease-driven T-cell signaling pathways in animal models. The company expects to advance this program into clinical trials in the first half of 2027. The appointment of Dr. Rahul Patel, an experienced rheumatologist and drug developer, will provide critical support in the strategic clinical advancement of Fortitude's therapeutic programs.
- Fortitude's lead AxSpA program is expected to enter the clinic in the first half of 2027.
The players
Fortitude Biomedicines
A private biotechnology company headquartered in Waltham, Massachusetts, pioneering next-generation immune cell targeting biologics and an antibody-drug conjugate (ADC) platform.
Dr. Rahul Patel
A board-certified rheumatologist who has joined Fortitude as the Senior Vice President of Clinical Development to accelerate the clinical development of the company's therapeutic programs.
Dr. Jesse Chen
The President and CEO of Fortitude Biomedicines.
What they’re saying
“With more than 50 percent of AxSpA patients unable to achieve adequate disease control, there continues to be a large unmet need in management of this disease. We are excited with the promising preclinical activity seen with our lead program.”
— Dr. Jesse Chen, President and CEO of Fortitude
“I am pleased to join Fortitude, to coordinate with exceptional team members and to advance an innovative pipeline of therapeutics. There remains a significant unmet need in rheumatology, including in AxSpA, for therapies that can advance the field further and potentially offer a higher bar of efficacy. I look forward to working with the Fortitude team as we advance to a clinical stage biotech ahead.”
— Dr. Rahul Patel, Senior Vice President of Clinical Development
What’s next
Fortitude's lead AxSpA program is expected to enter clinical trials in the first half of 2027.
The takeaway
Fortitude's new T-cell targeting bispecific antibody for AxSpA represents a promising advancement in addressing the significant unmet medical need for effective treatments that can improve outcomes and quality of life for the millions of patients suffering from this debilitating inflammatory disease.