- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
New Embolic Protection Device Proves Noninferior to Existing Option
TAVR Embolic Protection Devices Go Head to Head: PROTECT H2H
Apr. 2, 2026 at 12:23pm
Got story updates? Submit your updates here. ›
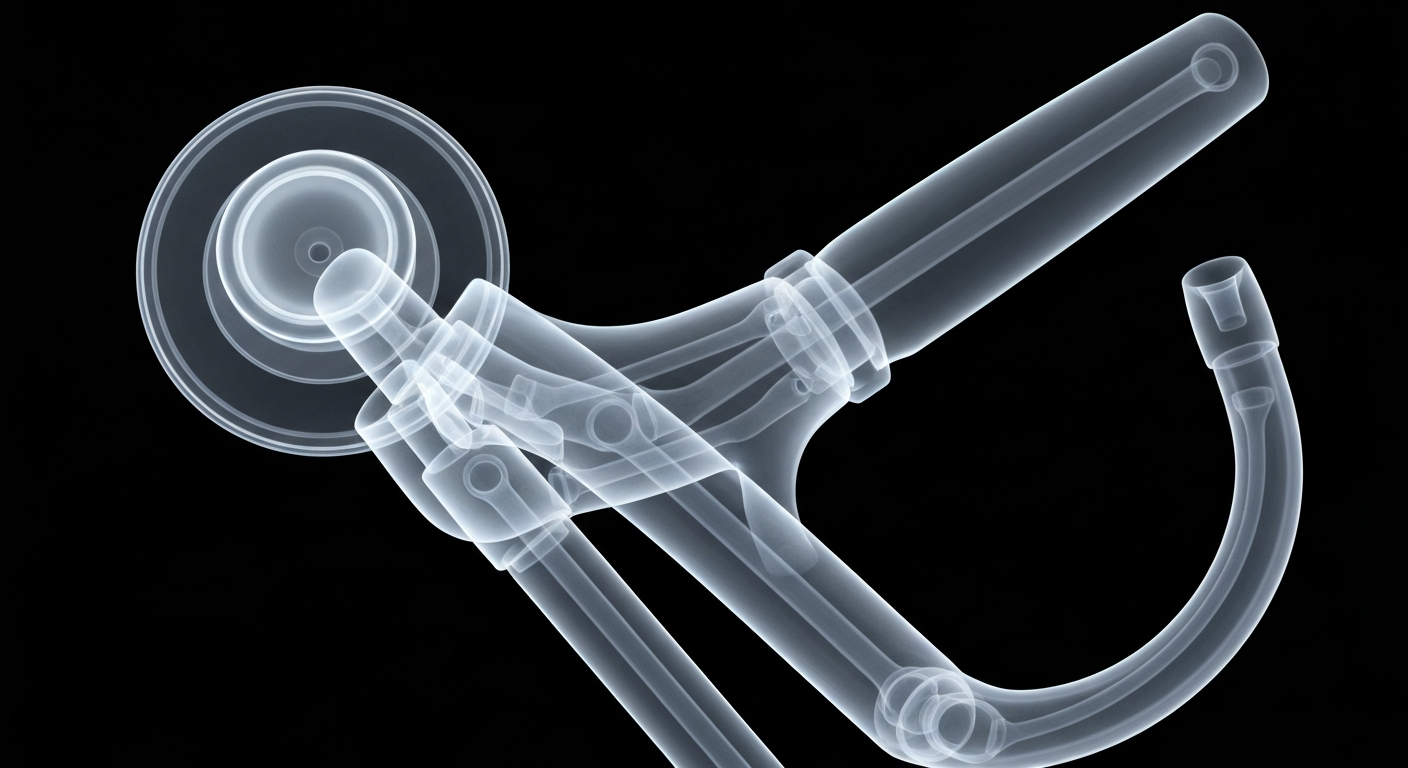
A new embolic protection device designed to capture more procedural debris during transcatheter aortic valve replacement (TAVR) was found noninferior to an existing FDA-approved device in a large randomized trial. While the new device was significantly better at capturing debris, the clinical benefits in terms of reducing stroke and other adverse events were less clear, highlighting the ongoing debate around the value of these protection devices.
Why it matters
Embolic protection devices are designed to capture debris released during TAVR procedures and prevent it from traveling to the brain and causing strokes. However, the clinical benefit of these devices has been hotly debated, as recent large trials have failed to show a significant reduction in stroke rates. This new study provides data on a next-generation device that appears better at debris capture, but the implications for patient outcomes remain uncertain.
The details
The randomized PROTECT H2H trial compared the new Emboliner Embolic Protection System to the FDA-approved Sentinel Cerebral Protection System. The primary endpoint of death, stroke, or acute kidney injury at 30 days was similar between the two groups, meeting the criteria for noninferiority. However, the new Emboliner device captured significantly more procedural debris, with a mean of 93 particles ≥ 150 μm compared to 31 for the Sentinel. Despite this, there was no statistically significant difference in stroke rates between the two arms. Procedural times were also longer with the newer Emboliner device, likely due to lack of prior experience compared to the Sentinel.
- The PROTECT H2H trial results were presented at the American College of Cardiology (ACC) Scientific Session 2026.
The players
Adam B. Greenbaum
A co-director of the Structural Heart and Valve Center at Emory University School of Medicine in Atlanta, and the lead investigator of the PROTECT H2H trial.
Michael N. Young
The director of the Structural Heart Disease Program at Dartmouth University's Geisel School of Medicine in Hanover, New Hampshire, and the discussant for the PROTECT H2H trial.
Emboliner Embolic Protection System
A new embolic protection device designed to capture more procedural debris during TAVR procedures.
Sentinel Cerebral Protection System
The only embolic protection device currently approved by the FDA for use during TAVR procedures.
What they’re saying
“Current protection devices only partially protect the cerebral circulation,”
— Adam B. Greenbaum, Co-director of the Structural Heart and Valve Center at Emory University School of Medicine
“The fact that the study associated the newer device with significantly greater debris capture without showing significantly greater protection against events, particularly stroke, which Young described as 'the holy grail' for proving the value of embolic protection devices, underscores the essence of the debate about these devices.”
— Michael N. Young, Director of the Structural Heart Disease Program at Dartmouth University's Geisel School of Medicine
What’s next
Researchers and clinicians will continue to closely monitor the performance and clinical benefits of embolic protection devices used during TAVR procedures. Further studies may be needed to definitively establish the value of these devices in improving patient outcomes.
The takeaway
While the new Emboliner device demonstrated superior debris capture compared to the existing Sentinel device, the lack of a clear clinical benefit in terms of reduced stroke and other adverse events highlights the ongoing uncertainty around the role of embolic protection in TAVR. Clinicians and patients will need to weigh the potential benefits against the added procedural complexity and cost of these devices.
New Orleans top stories
New Orleans events
Apr. 2, 2026
Comedy GumbeauxApr. 2, 2026
Scarlet Reverie & Hypno NunApr. 2, 2026
Brad Walker Quartet with Stanton Moore