- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
NX Development Corp Submits Supplemental New Drug Application to FDA for MEN-301 Study
The company's latest submission aims to expand the use of its Gleolan® product for fluorescence-guided surgery.
Apr. 6, 2026 at 10:50pm
Got story updates? Submit your updates here. ›
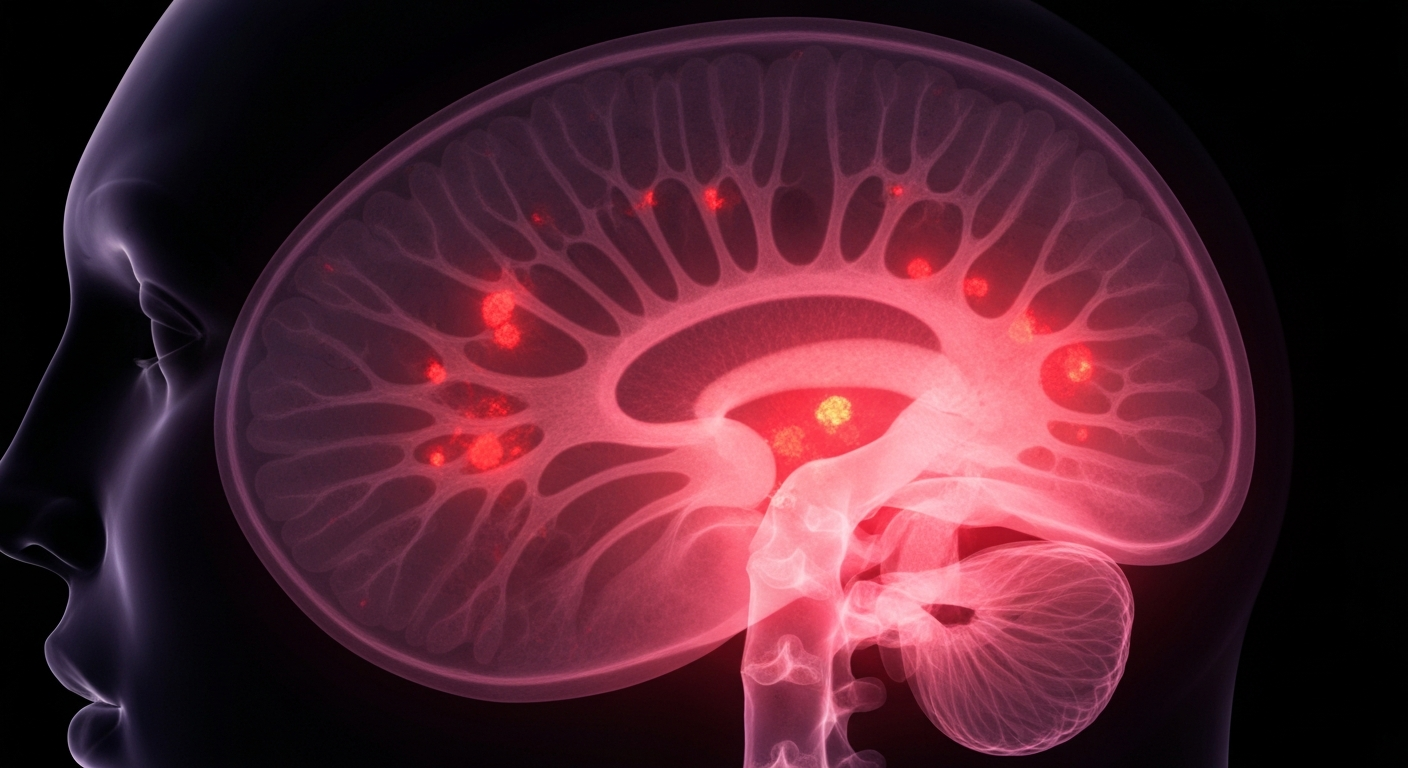 A groundbreaking new fluorescent dye technology is helping neurosurgeons better visualize and remove deadly brain tumors during delicate operations.Lexington Today
A groundbreaking new fluorescent dye technology is helping neurosurgeons better visualize and remove deadly brain tumors during delicate operations.Lexington TodayNX Development Corp (NXDC) has announced that it has submitted a supplemental new drug application to the FDA for its MEN-301 Study, which evaluated the use of the company's Gleolan® (aminolevulinic acid HCl) product to enhance fluorescence-guided surgery. This marks the first supplemental data submission to the FDA since Gleolan® was originally approved for high-grade glioma in 2017.
Why it matters
Fluorescence-guided surgery is an important tool for improving outcomes in complex procedures like brain tumor removal. NXDC's efforts to expand the approved uses of Gleolan® could lead to more patients benefiting from this technology and help advance the science of image-guided surgery.
The details
The MEN-301 Study was a clinical trial that analyzed the use of Gleolan® to aid in the visualization of tumor tissue during surgery. The successful completion of this study has allowed NXDC to submit supplemental data to the FDA, with the goal of expanding the approved indications for Gleolan® beyond its current use for high-grade glioma.
- NXDC originally received FDA approval for Gleolan® in 2017 for use in high-grade glioma procedures.
- The MEN-301 Study data analysis was recently completed in April 2026.
The players
NX Development Corp (NXDC)
A pharmaceutical company focused on developing and commercializing products for fluorescence-guided surgery, including the Gleolan® product.
What’s next
The FDA will now review NXDC's supplemental new drug application for the expanded use of Gleolan® based on the MEN-301 Study data.
The takeaway
NXDC's efforts to broaden the approved applications of its Gleolan® product demonstrate the company's commitment to advancing fluorescence-guided surgery techniques, which have the potential to improve outcomes for patients undergoing complex procedures like brain tumor removal.
Lexington top stories
Lexington events
Apr. 8, 2026
Keeneland Spring Race Meet




