- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
Pomona Today
By the People, for the People
Major Retailers Recall Over 3 Million Eye Drops Due to Sterility Concerns
FDA cites lack of assurance of sterility in products sold at CVS, Walgreens, and Rite Aid
Apr. 3, 2026 at 5:18pm
Got story updates? Submit your updates here. ›
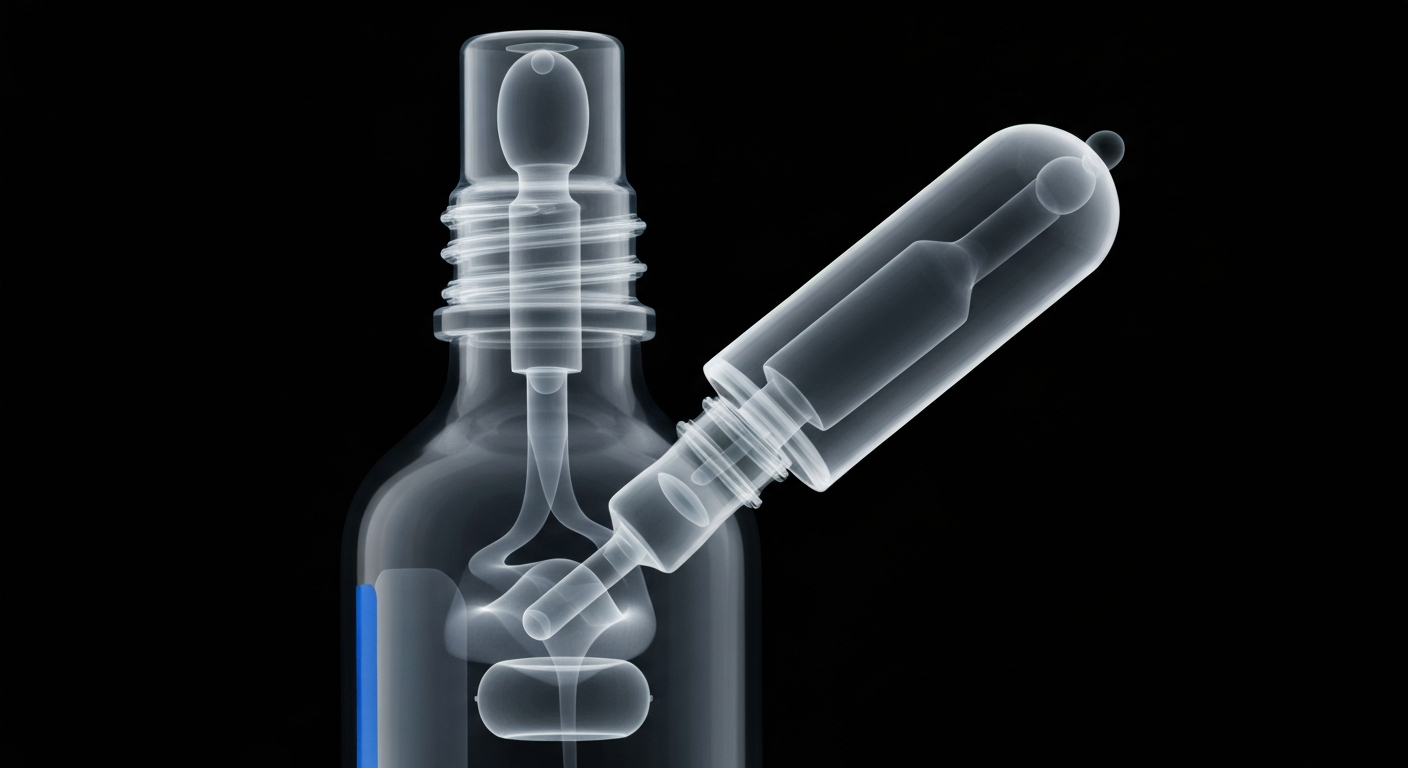
 An X-ray view exposes the internal mechanisms of a recalled eye drop bottle, highlighting the critical need for strict sterility standards in over-the-counter pharmaceuticals.Pomona Today
An X-ray view exposes the internal mechanisms of a recalled eye drop bottle, highlighting the critical need for strict sterility standards in over-the-counter pharmaceuticals.Pomona TodayA California pharmaceutical company has issued a voluntary recall of over 3 million bottles of over-the-counter eye drop products sold at major retailers like CVS, Walgreens, and Rite Aid. The Food and Drug Administration cited a 'lack of assurance of sterility' in the products, which are sold under brand names like 'Dry Eye Relief Eye Drops' and 'Artificial Tears Sterile Lubricant Eye Drops.' There have been no reports of injuries associated with the recalled items so far.
Why it matters
Eye drops are a common medication used by millions of Americans, so a large-scale recall of these products raises concerns about quality control and consumer safety. The FDA's classification of this as a 'Class II' recall indicates the potential for temporary or reversible health issues, underscoring the importance of addressing the sterility issues promptly.
The details
K.C. Pharmaceuticals, a company based in Pomona, California, manufactures the eye drop products that are being recalled. The recall covers 3,111,072 bottles of various eye drop brands sold at major drugstore chains. The FDA notice did not specify what exactly triggered the recall, but cited a 'lack of assurance of sterility' in the products.
- The recall began in early March 2026.
The players
K.C. Pharmaceuticals
A pharmaceutical company based in Pomona, California that manufactures the recalled eye drop products.
Food and Drug Administration (FDA)
The federal agency that cited the sterility issues and classified the recall as 'Class II.'
CVS
A major drugstore chain that sold the recalled eye drop products.
Walgreens
A major drugstore chain that sold the recalled eye drop products.
Rite Aid
A major drugstore chain that sold the recalled eye drop products.
What’s next
The FDA and K.C. Pharmaceuticals have not provided any timeline for when the sterility issues will be resolved or when the recalled products may return to store shelves.
The takeaway
This recall highlights the importance of strict quality control and sterility assurance in the manufacturing of over-the-counter medications like eye drops, which are widely used by consumers. It also underscores the need for vigilant monitoring and swift action by regulatory agencies like the FDA to protect public health.