- Today
- Holidays
- Birthdays
- Reminders
- Cities
- Atlanta
- Austin
- Baltimore
- Berwyn
- Beverly Hills
- Birmingham
- Boston
- Brooklyn
- Buffalo
- Charlotte
- Chicago
- Cincinnati
- Cleveland
- Columbus
- Dallas
- Denver
- Detroit
- Fort Worth
- Houston
- Indianapolis
- Knoxville
- Las Vegas
- Los Angeles
- Louisville
- Madison
- Memphis
- Miami
- Milwaukee
- Minneapolis
- Nashville
- New Orleans
- New York
- Omaha
- Orlando
- Philadelphia
- Phoenix
- Pittsburgh
- Portland
- Raleigh
- Richmond
- Rutherford
- Sacramento
- Salt Lake City
- San Antonio
- San Diego
- San Francisco
- San Jose
- Seattle
- Tampa
- Tucson
- Washington
Pioneering Spine Surgeons Perform First Case Using New FDA-Approved Synergy Disc
Drs. Todd Lanman and Joel Beckett complete three-level cervical disc replacement with motion-preserving technology.
Apr. 9, 2026 at 1:23am
Got story updates? Submit your updates here. ›
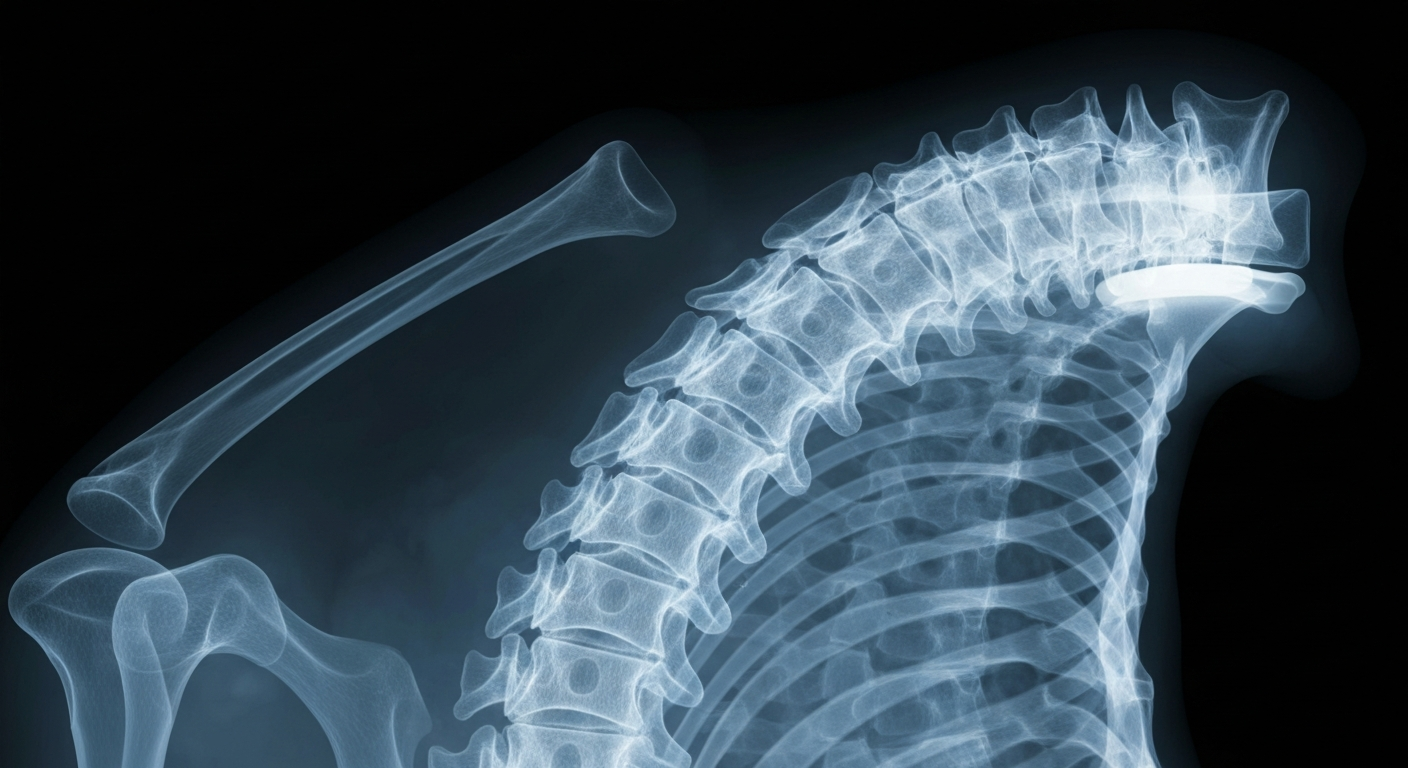 A pioneering spinal surgery utilizes cutting-edge technology to preserve motion and alignment, offering new hope for patients suffering from debilitating neck and arm pain.Beverly Hills Today
A pioneering spinal surgery utilizes cutting-edge technology to preserve motion and alignment, offering new hope for patients suffering from debilitating neck and arm pain.Beverly Hills TodayBeverly Hills spine surgeons Todd H. Lanman, MD and Joel S. Beckett, MD have performed the first post-approval case using the newly FDA-approved Synergy Disc®, a motion-preserving cervical disc replacement device. The procedure was completed on a 47-year-old patient with spinal cord compression and severe nerve root issues. This milestone case marks the clinical adoption of the Synergy Disc, which was recently granted Premarket Approval by the FDA for single-level cervical disc replacement.
Why it matters
The Synergy Disc is designed to not only preserve spinal motion but also support proper spinal alignment, which are key considerations in treating complex cervical degenerative conditions. This first successful case demonstrates the potential of this new technology to advance the standard of care for patients suffering from neck and arm pain due to cervical disc issues.
The details
The patient underwent a three-level cervical disc replacement procedure to decompress the spinal cord, relieve nerve compression, and remove bone spurs. Drs. Lanman and Beckett, who are pioneers in motion-preserving spine surgery, emphasized that the Synergy Disc's ability to maintain alignment while allowing controlled motion is an important advancement. The patient is recovering well with early reduction in pain, and neurologic recovery will be monitored over time.
- The procedure was performed on Monday, April 8, 2026.
- The Synergy Disc received FDA Premarket Approval five weeks prior, on March 4, 2026.
The players
Todd H. Lanman, MD
A spinal neurosurgeon with over three decades of experience who has practiced in Beverly Hills for the past 25 years. Dr. Lanman has also undergone 11 spinal surgeries himself, giving him a unique patient perspective.
Joel S. Beckett, MD
A board-certified, dual fellowship-trained neurosurgeon specializing in complex spinal reconstruction, artificial disc replacement, and motion-preserving spine surgery. Dr. Beckett has authored over 50 scientific publications and previously served as the Director of Neurosurgical Spinal Oncology at UCLA.
Synergy Spine Solutions
The company that developed the Synergy Disc, a motion-preserving artificial cervical disc that recently received FDA approval. Their vision is to identify and commercialize innovative spine technologies that improve patient outcomes.
What they’re saying
“This case reflects the continued evolution of motion-preserving spine surgery. The ability to address complex pathology while preserving motion is central to how we think about treating cervical degenerative disease.”
— Todd H. Lanman, MD, Spinal Neurosurgeon
“With the recent FDA approval of the Synergy Disc, we now have access to a technology designed not only to preserve motion but also to support segmental alignment. That combination is important as we continue to refine surgical decision-making in cervical spine care.”
— Joel S. Beckett, MD, Neurosurgeon
What’s next
The patient will be closely monitored for continued neurologic recovery as healing progresses following the successful three-level cervical disc replacement procedure. Broader clinical adoption of the Synergy Disc is expected as more surgeons gain experience with the newly approved technology.
The takeaway
This pioneering case showcases how the FDA-approved Synergy Disc technology can help advance the standard of care for patients with complex cervical spine issues. By preserving motion and supporting proper spinal alignment, this innovative device has the potential to improve outcomes and quality of life for those suffering from debilitating neck and arm pain.




